📋 Quick Summary
- Topic: Why leukemia compromises immunity, the most common infections in patients, and critical prevention strategies.
- Key Takeaway: Early recognition and medical consultation are critical for the best clinical outcomes.
- Remember: Always consult a qualified healthcare professional if you have concerns.
Infections, Leukemia, and Immune Health
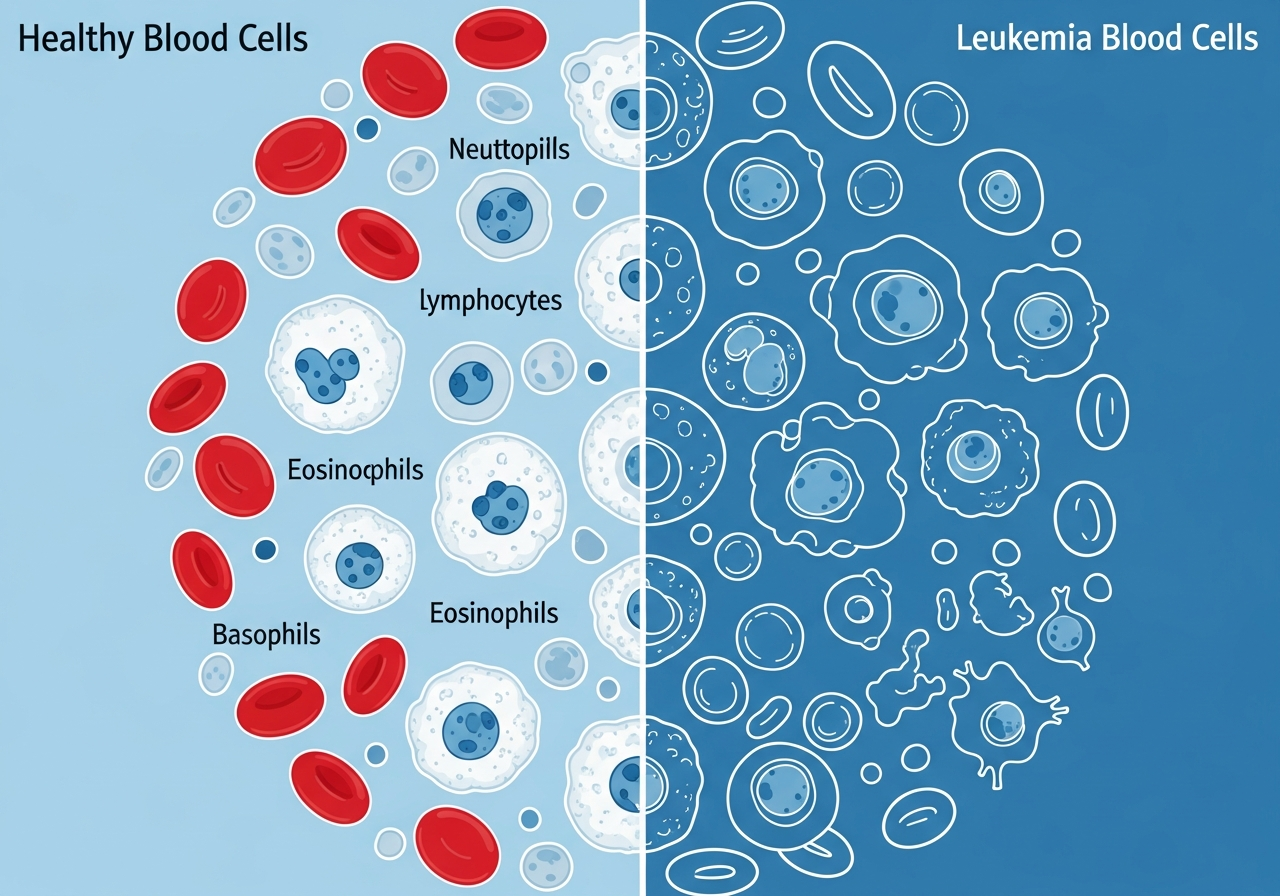
Infection is the leading cause of morbidity and mortality in leukemia patients. The immune system's ability to defend against bacteria, viruses, and fungi depends critically on the normal production and function of white blood cells — and leukemia disrupts this at its source, the bone marrow. The result is a state of profound immune compromise that makes even organisms normally harmless to healthy people potentially life-threatening to a leukemia patient. For a full overview of how infection fits among all leukemia symptoms, see the leukemia symptoms resource hub.
Understanding why leukemia patients are so vulnerable to infections, which infections are most dangerous, and what strategies most effectively prevent and treat them is essential knowledge for patients, families, and caregivers. Infections are not merely a complication of leukemia — they are frequently the immediate cause of death when leukemia is inadequately controlled. The risk varies meaningfully by leukemia type: patients with AML face rapid-onset neutropenia, while those with CLL contend with long-standing antibody deficiency.
The immune vulnerability seen in leukemia begins at symptom onset, before any treatment is given, and continues — often becoming most severe — during intensive chemotherapy. After remission is achieved, patients who undergo stem cell transplantation face another extended window of immune compromise requiring careful management. Effective management of infection risk is central to surviving leukemia, alongside treating the cancer itself.
Why Leukemia Severely Compromises the Immune System
The immune compromise in leukemia operates through multiple simultaneous mechanisms:
Neutropenia: The Primary Driver of Infection Risk
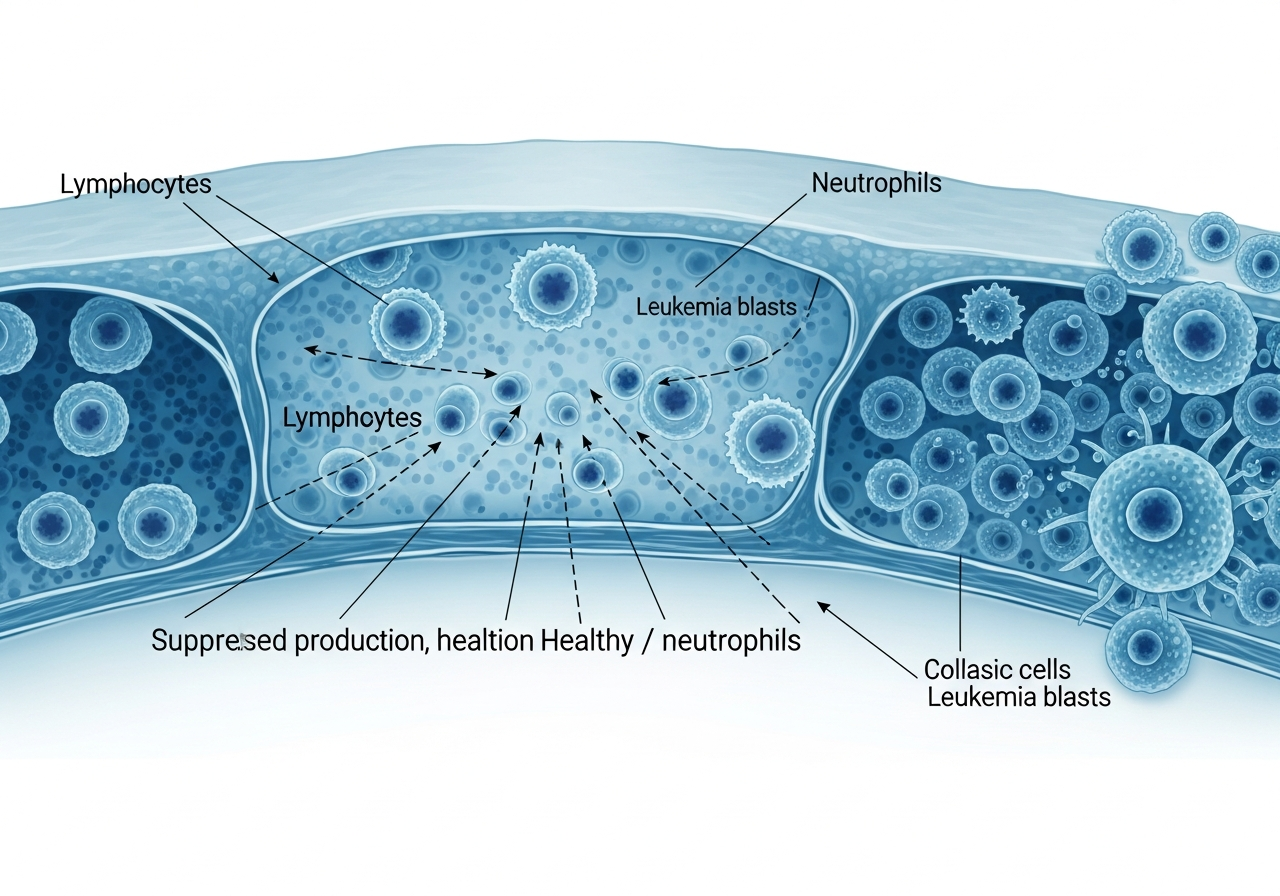
Neutrophils are the most abundant and most rapidly deployed white blood cells — the immune system's first responders to bacterial invasion. They engulf and destroy bacteria within minutes of contact. In leukemia, the proliferating leukemia cells crowd out normal neutrophil precursors in the bone marrow, severely reducing the neutrophil count (neutropenia). When the Absolute Neutrophil Count (ANC) falls below 500 cells/μL (normal is 1,500–8,000), the risk of serious bacterial infection increases dramatically. Below 100 cells/μL (profound neutropenia), the risk of life-threatening infection is almost certain without prophylactic antimicrobial measures.
Chemotherapy compounds this further — most cytotoxic agents directly suppress bone marrow function, creating a nadir (lowest point) in blood counts approximately 7–14 days after each treatment cycle.
Functional Immune Defects Beyond Neutrophil Count
In lymphocytic leukemias (particularly CLL), the leukemia cells are B lymphocytes — the cells responsible for producing antibodies (immunoglobulins). These leukemic B cells cannot produce functional antibodies, and their accumulation suppresses the production of normal B cells. The result is hypogammaglobulinemia — deficiency of protective antibodies — that leaves patients vulnerable to encapsulated bacteria (Streptococcus pneumoniae, Haemophilus influenzae) that healthy people normally resist through antibody-mediated immunity.
T-Cell Dysfunction
Both leukemia itself and its treatment (particularly with steroids, purine analogs like fludarabine, and CAR-T therapy) impair T-cell function. T cells are essential for controlling viral infections and fungal pathogens. T-cell deficiency dramatically increases vulnerability to viruses like cytomegalovirus (CMV), Epstein-Barr virus (EBV), and varicella-zoster virus (VZV), and to fungi like Aspergillus and Candida.
Physical Barriers
Mucositis — inflammation and ulceration of the gastrointestinal mucosa caused by chemotherapy — disrupts the physical barrier that normally prevents gut bacteria from entering the bloodstream. This creates a direct route for bacterial translocation and is a significant contributor to bloodstream infections (bacteremia) during treatment.
Most Common and Dangerous Infections in Leukemia
| Type | Common Pathogens | Clinical Presentation | Risk Period |
|---|---|---|---|
| Bacterial | E. coli, Klebsiella, Pseudomonas, Staphylococcus | Fever, sepsis, pneumonia, UTI | Neutropenic nadir (7–14 days post-chemo) |
| Fungal | Aspergillus, Candida, Pneumocystis jirovecii | Pneumonia, bloodstream infection, sinusitis | Prolonged neutropenia; post-transplant |
| Viral | CMV, EBV, VZV, HSV, RSV, influenza | Pneumonitis, encephalitis, colitis, hepatitis | T-cell depleted phases; post-transplant |
| Encapsulated bacteria | S. pneumoniae, H. influenzae | Pneumonia, meningitis, sepsis | CLL with hypogammaglobulinemia |
Febrile Neutropenia: The Most Critical Emergency
Febrile neutropenia — defined as a fever above 38.3°C (101°F) or sustained above 38°C (100.4°F), combined with an ANC below 500 cells/μL — is a medical emergency requiring immediate hospital evaluation and empiric broad-spectrum antibiotic treatment. Without prompt treatment, bacterial sepsis can be fatal within hours. Any leukemia patient who develops fever while neutropenic must go to the emergency room immediately — not wait for a morning clinic appointment.
Factors That Increase Infection Risk
Several factors compound the baseline immune compromise of leukemia:
- Degree and duration of neutropenia: ANC below 100 cells/μL for more than 7 days (prolonged profound neutropenia) carries the highest risk for serious fungal and bacterial infection.
- Central venous catheters (CVCs): Hickman lines, PICC lines, and port-a-caths provide essential venous access but also create a direct infection entry point for skin bacteria.
- Corticosteroids: Widely used in leukemia treatment regimens, steroids suppress both innate and adaptive immunity, particularly increasing fungal and viral risk.
- Mucositis: Oral and gastrointestinal ulceration from chemotherapy allows gut bacteria to enter the bloodstream.
- Allogeneic stem cell transplant: The most immunosuppressive treatment modality — patients are profoundly immunocompromised for 6–12 months or longer, with specific vulnerability windows for different pathogens.
- Purine analogs (fludarabine, cladribine): These chemotherapy agents produce profound, long-lasting T-cell depletion beyond the neutropenic nadir.
Diagnostic Approach to Suspected Infection in Leukemia
When a leukemia patient develops fever or other infection signs, a rapid, systematic evaluation is essential. Standard workup includes:
- Blood cultures (from peripheral blood and from each CVC lumen) before antibiotics are started
- Complete blood count to assess ANC and confirm neutropenia
- Chest X-ray or CT (if X-ray is non-diagnostic); pulmonary aspergillosis may require high-resolution CT
- Urinalysis and urine culture
- Nasal/throat swabs for viral respiratory pathogens
- Serum galactomannan and beta-D-glucan (fungal biomarkers)
- CMV and EBV PCR (in post-transplant patients or those on purine analogs)
The urgency of this workup cannot be overstated. In a neutropenic patient, antibiotics should be started within 60 minutes of triage — earlier if the patient is hemodynamically unstable. The CBC and differential are fundamental, and the hematology team should be contacted immediately when infection is suspected in a leukemia patient.
Treatment and Prevention of Infections in Leukemia
Antimicrobial Prophylaxis
Prophylactic (preventive) antimicrobial agents are routinely given during high-risk periods:
- Antibacterial prophylaxis: Fluoroquinolones (e.g., levofloxacin) during the expected neutropenic nadir substantially reduce bacterial infections in high-risk patients.
- Antifungal prophylaxis: Azoles (fluconazole, posaconazole, or voriconazole) prevent Candida and Aspergillus infections during neutropenia and after transplant.
- Anti-pneumocystis prophylaxis: Trimethoprim-sulfamethoxazole (TMP-SMX) prevents Pneumocystis jirovecii pneumonia in patients on prolonged immunosuppression.
- Antiviral prophylaxis: Acyclovir or valacyclovir prevents herpes simplex and varicella-zoster reactivation; antiviral agents are also used for CMV prevention after transplant.
Granulocyte Colony-Stimulating Factor (G-CSF)
G-CSF (filgrastim, pegfilgrastim) stimulates neutrophil production in the bone marrow. Its use after chemotherapy shortens the duration of neutropenia and reduces the risk and severity of febrile neutropenia. It is a standard component of most intensive chemotherapy regimens.
Intravenous Immunoglobulin (IVIG)
In CLL patients with hypogammaglobulinemia and recurrent serious bacterial infections, IVIG infusions can supplement deficient antibody levels and reduce infection frequency.
Vaccination
Leukemia patients should receive influenza vaccination annually and other non-live vaccines as recommended by their oncologist. Live vaccines (measles, varicella, yellow fever) are generally contraindicated during active treatment and significant immunosuppression.
Prognosis and Infection Management
The prognosis of infection in leukemia has improved significantly with modern prophylactic strategies, rapid diagnostic tools, and broad-spectrum antimicrobial therapies. Febrile neutropenia that is promptly treated has a mortality rate of well under 5% in most risk categories — though fungal pneumonia, particularly aspergillosis, remains a serious and potentially fatal complication despite antifungal treatment, particularly in patients with prolonged neutropenia. Overall survival in leukemia is increasingly determined by how effectively infection risk is managed across the full treatment journey — from initial diagnosis through treatment selection and into long-term survivorship planning. Understanding your specific leukemia test results, including ANC trends, is a practical tool for infection risk awareness.
Infection Prevention at Home
Patients can take meaningful action to reduce infection risk during treatment:
- Hand hygiene: Frequent, thorough handwashing with soap and water — or alcohol-based hand gel — is the single most effective infection prevention measure.
- Avoid sick contacts: Avoid close contact with anyone who has respiratory illness, chickenpox, or other contagious infections during periods of neutropenia.
- Food safety: Avoid raw or undercooked meats, raw eggs, unpasteurized dairy, and high-risk foods (sushi, deli meats, soft cheeses) during neutropenic periods.
- Oral hygiene: Rinse with salt water or prescribed mouthwash multiple times daily to reduce oral bacteria and prevent mucositis-related infections.
- Skin care: Keep skin clean and moisturized; inspect CVC site daily for redness, swelling, or discharge.
- Avoid crowds and poorly ventilated spaces: High-traffic indoor environments increase exposure to airborne pathogens.
Caregiver Guidance
Caregivers are the first line of defense against infection in a leukemia patient's home. Caregivers should maintain their own vaccinations — particularly annual influenza vaccination — to reduce the risk of transmitting preventable infections. Good hand hygiene before preparing food, before and after patient contact, and after being in public spaces is essential. Caregivers should also monitor the patient for early signs of infection: subtle fever, unusual chills, redness or swelling at any wound or catheter site, or behavioral change (confusion, unusual lethargy). Early recognition enables faster treatment, which saves lives.
When to Seek Urgent Medical Care
Any leukemia patient — particularly during active treatment — who develops a fever above 38.3°C (101°F) or 38°C (100.4°F) sustained over one hour, should seek emergency evaluation immediately, without waiting. Do not give fever-reducing medication and observe — seek care first. Additionally, seek emergency care for:
- Rigors (uncontrolled shaking chills)
- Rapid heart rate, low blood pressure, or confusion alongside fever
- Redness, warmth, swelling, or discharge around a central venous catheter
- New severe mouth sores or difficulty swallowing
- New productive cough with fever
- Blood in urine, stool, or vomit
🚨 Go to the Emergency Room Immediately If:
- Fever above 101°F (38.3°C) with known low white blood cell count — febrile neutropenia is an emergency
- Rigors, rapid heartbeat, and low blood pressure — possible sepsis
- Redness or discharge around a central line or port
- Confusion, severe weakness, or sudden change in condition
💬 Questions to Ask Your Healthcare Team
- What is my current ANC, and am I at high risk for febrile neutropenia?
- What antimicrobial prophylaxis am I on, and what does each drug prevent?
- At what temperature should I go directly to the emergency room?
- Should I have G-CSF injections to shorten my neutropenic period?
- Are there food restrictions I should follow during neutropenia?
- What infections am I most at risk for based on my specific leukemia and treatment?
This content is for informational and educational purposes only. It is not a substitute for professional medical advice, diagnosis, or treatment. Always consult your physician or a qualified healthcare provider with any questions about a medical condition. Never disregard professional medical advice or delay seeking it because of information you have read on this website. Read our full disclaimer.
Frequently Asked Questions
Leukemia cells crowd out healthy white blood cells in the bone marrow, leaving the body without its primary defense against pathogens.
Pneumonia, sepsis, and severe fungal infections (like aspergillosis or candidemia) are particularly life-threatening.
Frequent handwashing, avoiding sick individuals, food safety practices, and following a doctor's guidance on preventive antibiotics or antivirals.
