
💊 Leukemia Treatment Options — Complete Guide
A complete, medically accurate guide to every leukemia treatment modality — from standard chemotherapy phases to precision targeted drugs, immunotherapy, stem cell transplantation, and cutting-edge clinical trials.
The Principles of Leukemia Treatment
Leukemia treatment has undergone a revolutionary transformation over the past three decades. Where once chemotherapy was the only available option — applied with limited precision and significant toxicity — modern oncology now delivers treatment through multiple complementary modalities, each targeting a specific aspect of leukemia biology. The treatment a patient receives in 2025 is increasingly personalized to their specific leukemia type, molecular profile, age, overall fitness, and treatment goals.
Treatment decisions begin with a complete understanding of the leukemia's molecular biology, obtained through comprehensive bone marrow biopsy and molecular testing. The specific genetic mutations and chromosomal abnormalities present in the leukemia cells determine which targeted drugs are active, what risk category the patient falls into, and whether stem cell transplantation is recommended. Treatment is then delivered by a multidisciplinary team that typically includes hematologist-oncologists, advanced practice providers, pharmacists, nurses, social workers, and psychologists.
The goal of treatment — whether cure, long-term remission, or disease control — depends on the specific leukemia type. Acute leukemias are treated with curative intent: the aim is to eliminate all detectable leukemia from the body and prevent relapse. Chronic leukemias like CLL and CML are more commonly managed with the goal of controlling the disease long-term while maintaining quality of life, rather than achieving cure in all patients.
Treatment Modalities by Leukemia Type
| Leukemia Type | First-Line Treatment | Key Targeted Options | Transplant Role |
|---|---|---|---|
| Childhood ALL | Multi-agent intensive chemo (2.5–3 years) | TKI (Ph+ ALL); blinatumomab; inotuzumab | Very high-risk or relapsed only |
| Adult ALL | Intensive chemo ± TKI (if Ph+) | Ponatinib, dasatinib (Ph+); CAR-T (relapsed) | Recommended for most high-risk patients |
| AML (standard risk) | Induction chemo (7+3 or variants) | Midostaurin (FLT3+); ATRA+ATO (APL) | Adverse risk or relapse |
| CLL | Watch-and-wait (early) or BTKi / venetoclax | Ibrutinib, acalabrutinib, venetoclax+obinutuzumab | Rare; selected relapsed/refractory cases |
| CML (chronic phase) | Tyrosine kinase inhibitor (TKI) | Imatinib, dasatinib, nilotinib, ponatinib (T315I) | Blast crisis or TKI-resistant only |
Understanding Chemotherapy Phases
For acute leukemias, chemotherapy is delivered in structured phases, each with a distinct goal. Understanding these phases helps patients and families know what to expect across the months-long treatment journey:
- Induction (Weeks 1–8): Intensive, often in-hospital chemotherapy aimed at achieving complete remission — reducing leukemia cells in the bone marrow to undetectable levels and restoring normal blood counts. Most patients achieve remission after induction.
- Consolidation (Months 2–8): Additional chemotherapy cycles to eliminate residual disease (minimal residual disease, MRD) that survives induction. May include different drug combinations, high-dose regimens, or stem cell transplantation in eligible patients.
- Maintenance (Months 8–36+): Lower-intensity oral chemotherapy delivered over months to years to prevent relapse. Critical in ALL; not used in most AML protocols. Allows patients to return to near-normal activity while protecting against disease recurrence.
- CNS-directed therapy: In ALL, leukemia cells can hide in the central nervous system — specialized intrathecal (spinal) chemotherapy throughout all phases prevents CNS relapse.
All Treatment Articles — Complete Library
Leukemia Treatment Options Explained
A complete overview of all treatment modalities, decision factors, and the multidisciplinary care team approach.
Read more →
Chemotherapy for Leukemia
Induction, consolidation, and maintenance phases, common drugs, side effects, and what patients should expect.
Read more →
Targeted Therapy for Leukemia
TKIs for CML and ALL, BCL-2 inhibitors for CLL, IDH inhibitors for AML, and the expanding world of precision oncology.
Read more →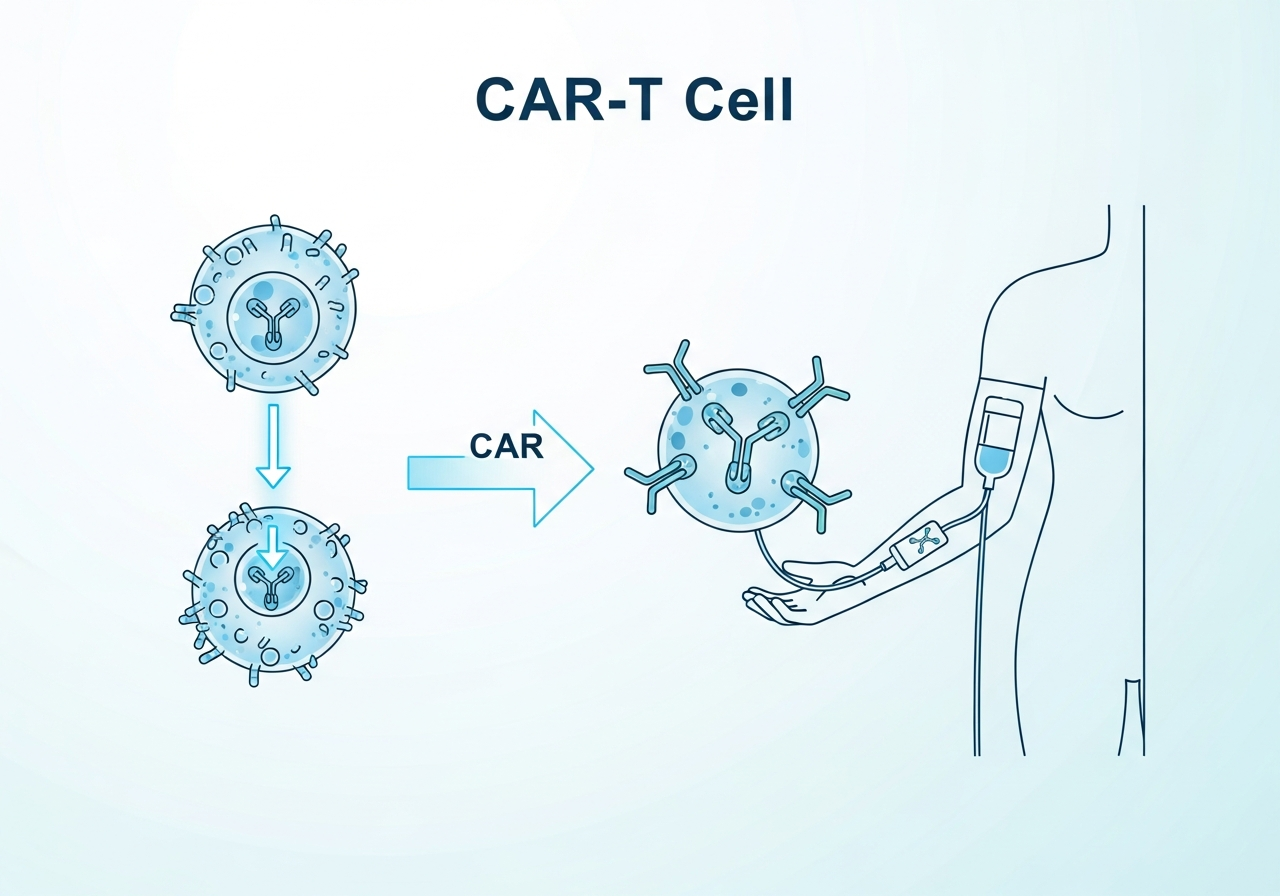
Immunotherapy for Leukemia
CAR-T cell therapy, bispecific antibodies, checkpoint inhibitors, and how these treatments harness the immune system.
Read more →
Stem Cell Transplants in Leukemia Recovery
Autologous vs. allogeneic transplants, donor matching, the recovery process, and the risks and benefits of this intensive therapy.
Read more →
Clinical Trials, Treatment Decisions, and Recovery Planning
How clinical trials work, how to find them, shared decision-making with oncologists, and long-term survivorship care.
Read more →Featured Guides
Leukemia Treatment Options Explained
The complete overview of all treatment modalities, the multidisciplinary care team, how leukemia type and molecular profile drive treatment selection, and what patients should expect at each stage.
Read full guide →Targeted Therapy for Leukemia
TKIs for CML and ALL, BCL-2 inhibitors for CLL, IDH and FLT3 inhibitors for AML — a deep dive into the expanding world of drugs that attack specific molecular drivers of leukemia.
Read full guide →Clinical Trials: Access to Tomorrow's Treatments
Clinical trials are the mechanism through which every currently approved leukemia treatment was developed — and they remain the most important pathway to improved outcomes for patients, particularly those with rare, high-risk, or relapsed disease. Participation in a clinical trial does not mean receiving inferior care; in most cases, trial participants receive at least the current standard of care plus access to an investigational agent that may prove superior. Major leukemia clinical trial groups — including the Children's Oncology Group (COG), ECOG-ACRIN, and the German Consortium — run hundreds of trials at any given time. Our guide to clinical trials and treatment decisions explains how to find and evaluate trial options, and what shared decision-making with your oncologist looks like.
Treatment Options — Frequently Asked Questions
Leukemia treatment is determined by the specific type, molecular subtype, patient age, and overall health. Options include multi-drug chemotherapy, oral targeted therapies (TKIs, BTK inhibitors, BCL-2 inhibitors), immunotherapy (CAR-T, bispecific antibodies), stem cell transplantation, and clinical trial participation. Some early-stage chronic leukemias (particularly CLL) may be managed initially with watchful waiting.
Targeted therapies are drugs designed to block specific molecular abnormalities that drive leukemia growth. Examples include tyrosine kinase inhibitors (imatinib, dasatinib) for BCR-ABL1 in CML and Ph+ ALL; FLT3 inhibitors (midostaurin, gilteritinib) for FLT3-mutated AML; IDH inhibitors (ivosidenib, enasidenib) for IDH-mutated AML; and venetoclax (BCL-2 inhibitor) for CLL and AML. They typically cause fewer side effects than broad chemotherapy.
CAR-T (chimeric antigen receptor T-cell) therapy involves collecting T cells from the patient's blood, genetically engineering them to express a receptor that targets a specific protein on leukemia cells (commonly CD19 or CD22 in B-cell ALL), and reinfusing them to attack the cancer. It is FDA-approved for relapsed/refractory B-cell ALL and has achieved remissions in patients who failed all other treatments. Major risks include Cytokine Release Syndrome (CRS) and neurological toxicity.
Allogeneic stem cell transplantation is generally recommended for patients with high-risk acute leukemias (adverse cytogenetics, certain molecular mutations), those who have relapsed, and some patients with CMML or high-risk MDS. It is the only potentially curative approach for some leukemia subtypes that respond poorly to chemotherapy alone. Patient age, fitness, and donor availability all factor into transplant eligibility.
Induction chemotherapy is the intensive initial treatment aimed at achieving complete remission — typically requiring hospitalization and producing significant blood count suppression over 4–8 weeks. Maintenance chemotherapy is a prolonged, lower-intensity phase (typically oral medications over 1–3 years, primarily in ALL) designed to prevent relapse by eliminating residual leukemia cells that survive induction and consolidation.
Yes — clinical trials are strongly recommended for all leukemia patients, particularly those with rare subtypes, high-risk disease, or relapse. Participation provides access to investigational agents that may be more effective than current standard therapies, and all trial participants receive at least the current standard of care. Search ClinicalTrials.gov or ask your oncologist about open trials at your center.
Explore Related Topics
This content is for informational and educational purposes only. It is not a substitute for professional medical advice, diagnosis, or treatment. Always consult your physician or a qualified healthcare provider with any questions about a medical condition. Read our full disclaimer.