📋 Quick Summary
- Topic: An overview of ALL, AML, CLL, CML, acute vs. chronic presentation, and lymphocytic vs. myeloid origins.
- Key Takeaway: Early recognition and medical consultation are critical for the best clinical outcomes.
- Remember: Always consult a qualified healthcare professional if you have concerns.
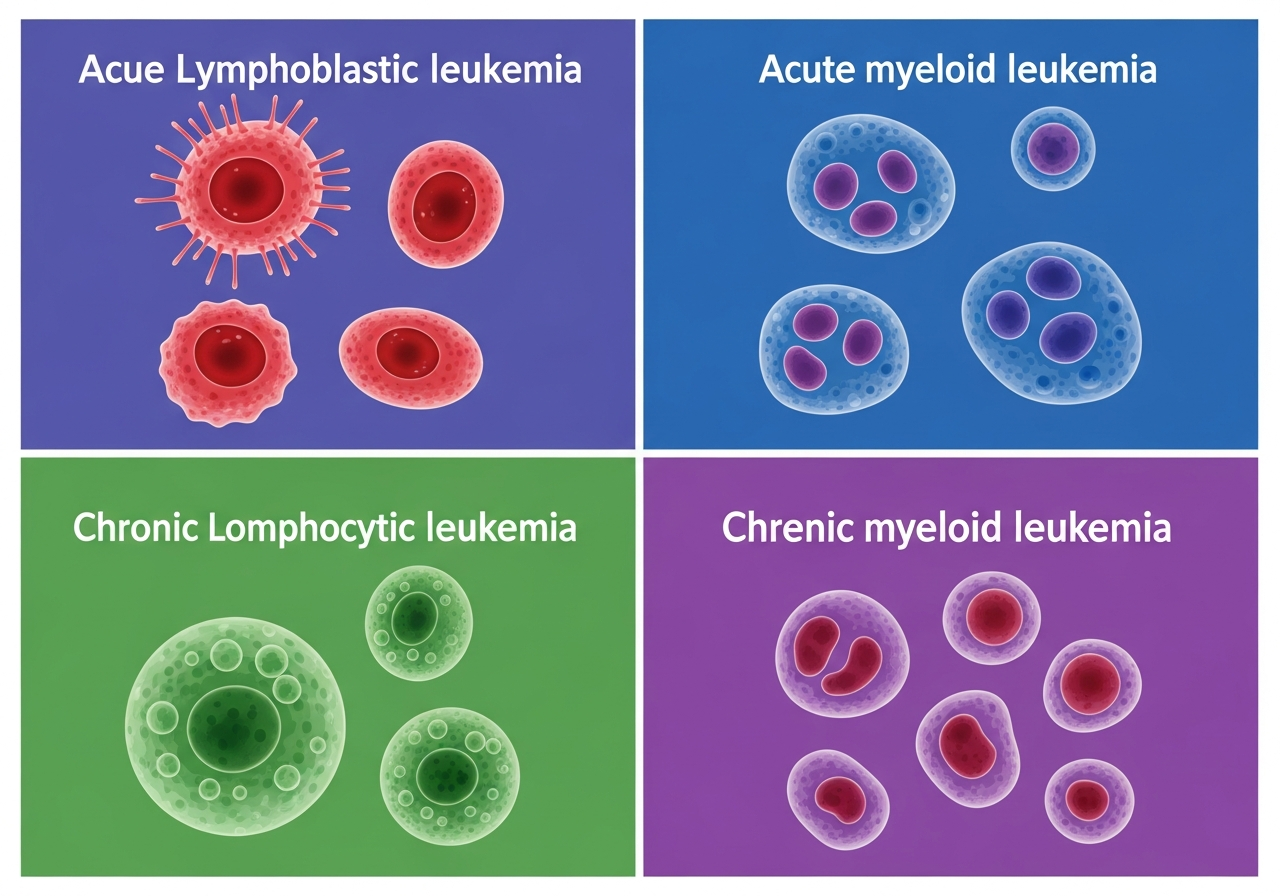
Types of Leukemia: An Essential Guide
Leukemia is not a single disease. It is a diverse family of blood cancers that share one fundamental characteristic — abnormal proliferation of blood cells originating in the bone marrow — but differ markedly in their biology, clinical course, treatment, and prognosis. Knowing the specific type of leukemia a patient has is the single most important factor in determining the appropriate treatment and predicting outcomes.
There are more than a dozen distinct leukemia types and subtypes, but four major categories account for the vast majority of diagnoses:
- Acute Lymphoblastic Leukemia (ALL)
- Acute Myeloid Leukemia (AML)
- Chronic Lymphocytic Leukemia (CLL)
- Chronic Myeloid Leukemia (CML)
Understanding what "acute" versus "chronic" means, what "lymphoblastic/lymphocytic" versus "myeloid" means, and how these distinctions translate into clinical behavior and treatment decisions provides an essential framework for patients and families navigating a leukemia diagnosis.
The Classification System: Acute vs. Chronic, Lymphoid vs. Myeloid
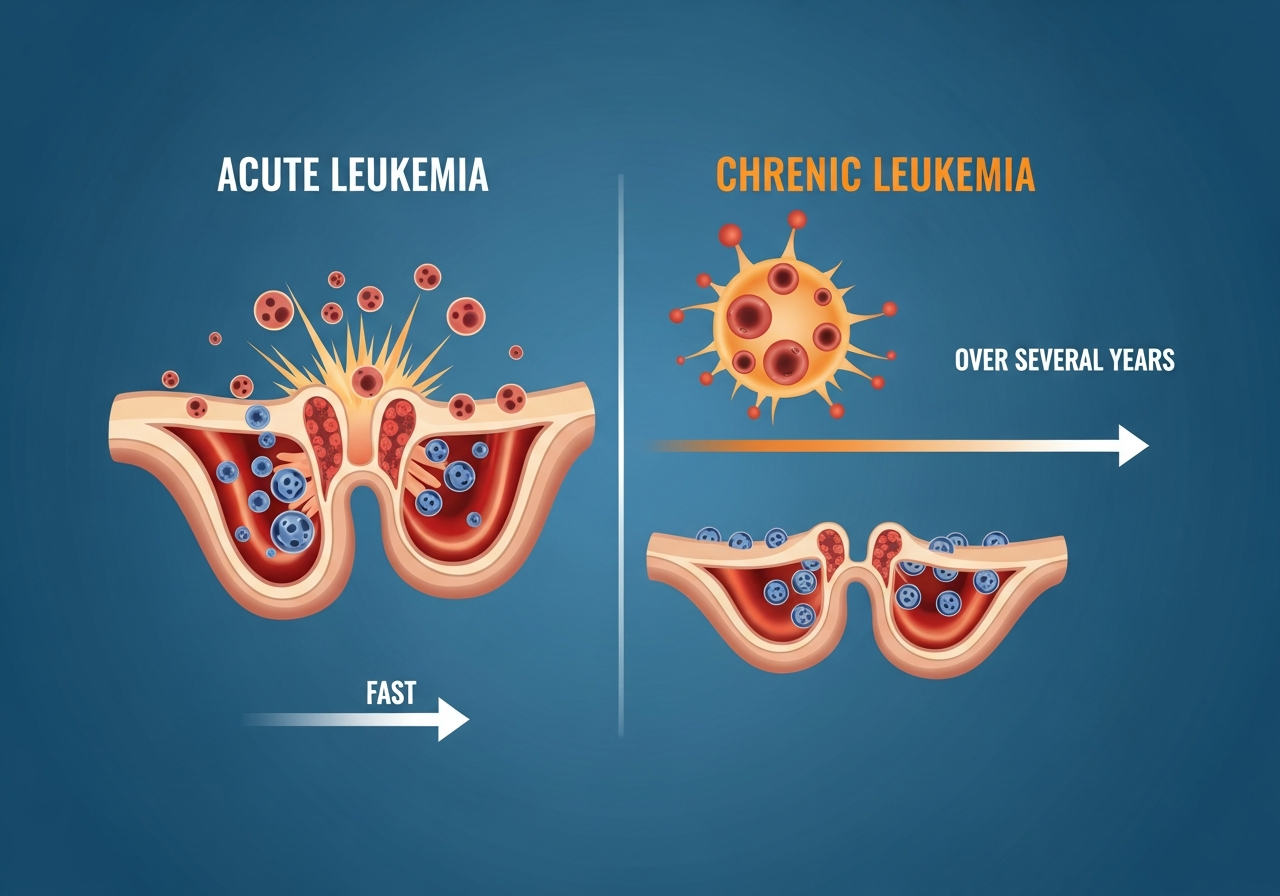
Acute vs. Chronic
The distinction between acute and chronic leukemia refers to the maturity of the malignant cells and the speed of disease progression — not to the severity of the condition in an absolute sense:
- Acute leukemia: The malignant cells are immature "blast" cells that have lost the ability to differentiate into mature, functional blood cells. They proliferate rapidly, and without treatment, acute leukemias can be fatal within weeks to months. Paradoxically, because acute leukemias involve rapidly dividing cells, they often respond dramatically — and potentially curatively — to intensive chemotherapy.
- Chronic leukemia: The malignant cells are more mature and retain partial function. They proliferate more slowly, and the disease typically progresses over months to years rather than weeks. Chronic leukemias are generally not curable with chemotherapy alone (with exceptions), but many patients live for years or decades with good quality of life on modern targeted therapies.
Lymphoid vs. Myeloid
This distinction refers to the type of blood cell precursor from which the leukemia originates:
- Lymphoid (lymphocytic): Originates from lymphoid stem cells — precursors of B lymphocytes, T lymphocytes, and NK cells. ALL and CLL are lymphoid leukemias.
- Myeloid: Originates from myeloid stem cells — precursors of red blood cells, platelets, granulocytes (neutrophils, eosinophils, basophils), and monocytes. AML, CML, and CMML are myeloid leukemias.
Acute Lymphoblastic Leukemia (ALL)
ALL is the most common leukemia of childhood, accounting for approximately 75% of all pediatric leukemia diagnoses and representing approximately 30% of all childhood cancers. In adults, ALL is less common but tends to have a more aggressive course and lower cure rates than in children.
ALL arises from malignant transformation of a lymphoid precursor cell (lymphoblast). The leukemia cells rapidly proliferate in the bone marrow, displacing normal hematopoiesis and producing the classic triad of anemia, thrombocytopenia, and neutropenia. ALL-specific features include prominent bone pain, lymphadenopathy, and in some subtypes (T-cell ALL), a mediastinal mass that can cause respiratory compromise.
ALL is further classified by cell lineage (B-cell vs. T-cell) and by molecular/cytogenetic features. The most important molecular marker is the Philadelphia chromosome (BCR-ABL fusion gene, present in ~5% of childhood ALL and ~25–30% of adult ALL), which indicates high-risk disease but also identifies a therapeutic target — tyrosine kinase inhibitors (TKIs) like dasatinib — that dramatically improve outcomes in Ph+ ALL.
Five-year survival in childhood ALL exceeds 90%. In adults, survival rates range from 40–60% depending on age and molecular risk category, with ongoing improvements from newer targeted agents. Read the full guide to ALL.
Acute Myeloid Leukemia (AML)
AML is the most common acute leukemia of adulthood, with median diagnosis age of approximately 68 years. It is characterized by the rapid expansion of immature myeloid blast cells that crowd out the bone marrow and suppress all normal blood cell production — producing profound anemia, thrombocytopenia (with significant bleeding risk), and neutropenia (with extreme infection susceptibility).
AML is biologically heterogeneous — it encompasses multiple subtypes (classified by the FAB and WHO systems) with distinct molecular profiles. Key molecular subtypes include:
- APL (Acute Promyelocytic Leukemia, AML-M3): Distinguished by the PML-RARA fusion gene; associated with severe DIC; highly treatable with ATRA + arsenic trioxide (cure rate ~90%)
- FLT3-mutated AML: High-risk; treated with FLT3 inhibitors (midostaurin, gilteritinib) in combination with chemotherapy
- IDH1/IDH2-mutated AML: Targetable with IDH inhibitors (enasidenib, ivosidenib)
- NPM1-mutated AML: Generally favorable prognosis with standard treatment
Overall five-year survival for AML is approximately 29%, with significant variation by age, molecular profile, and response to initial treatment. Allogeneic stem cell transplantation is the standard consolidation for eligible patients with high-risk or intermediate-risk disease. Read the full guide to AML.
Chronic Lymphocytic Leukemia (CLL)
CLL is the most common leukemia in adults in Western countries, diagnosed primarily in patients over 60 years of age. It is characterized by the accumulation of mature but non-functional B lymphocytes in the blood, bone marrow, lymph nodes, and spleen. Unlike acute leukemias, CLL typically has an indolent (slow-growing) course — many patients live for decades after diagnosis and may never require treatment.
CLL staging (Rai and Binet systems) is based on lymph node involvement, spleen/liver involvement, anemia, and thrombocytopenia. Early-stage patients (Rai 0; Binet A) without symptoms are typically managed with "watchful waiting" — active surveillance without treatment — until disease progression triggers treatment criteria.
The treatment landscape for CLL has been revolutionized by targeted agents:
- BTK inhibitors: Ibrutinib, acalabrutinib, zanubrutinib — oral daily medications that have transformed CLL treatment with high response rates and excellent tolerability
- BCL-2 inhibitors: Venetoclax — highly active, particularly in combination with anti-CD20 antibodies (obinutuzumab, rituximab)
- PI3K inhibitors: Idelalisib, duvelisib — less commonly used due to toxicity profile
CLL is not generally considered curable with current therapies, but many patients achieve deep, prolonged remissions. Median survival with modern therapy exceeds 10–15 years. Read the full guide to CLL.
Chronic Myeloid Leukemia (CML)
CML is defined by the presence of the Philadelphia chromosome — a translocation between chromosomes 9 and 22 that creates the BCR-ABL fusion gene. BCR-ABL encodes a constitutively active tyrosine kinase that drives uncontrolled myeloid cell proliferation. The discovery of imatinib (Gleevec) — the first tyrosine kinase inhibitor — has made CML one of the great success stories of targeted therapy in oncology.
CML has three clinical phases:
- Chronic phase: Mild to moderate symptoms; excellent response to TKIs; most patients diagnosed here
- Accelerated phase: Disease progression; less responsive to initial TKIs; may require second- or third-generation agents
- Blast phase (blast crisis): Resembles acute leukemia; aggressive; intensive treatment required
With TKI therapy (imatinib, dasatinib, nilotinib, bosutinib, ponatinib), the vast majority of CML patients in chronic phase achieve complete cytogenetic responses and remain in sustained remission. Median survival approaches that of the general population. A proportion of patients who achieve deep molecular remission can discontinue TKI therapy and maintain "treatment-free remission." Read the full guide to CML.
Rarer Leukemia Subtypes
Beyond the four main types, several rarer subtypes are clinically significant:
- CMML (Chronic Myelomonocytic Leukemia): A hybrid disorder with features of both myelodysplastic syndrome (MDS) and myeloproliferative neoplasm (MPN); characterized by monocytosis; treated with hypomethylating agents or transplant.
- Hairy Cell Leukemia (HCL): A rare B-cell leukemia with characteristic "hairy" cell projections; excellent prognosis with cladribine or vemurafenib (for BRAF-mutated HCL).
- Large Granular Lymphocytic (LGL) Leukemia: A chronic T-cell or NK-cell leukemia, often associated with autoimmune conditions; treated with immunosuppressants.
- Prolymphocytic Leukemia (PLL): A rare and aggressive lymphocytic leukemia requiring intensive treatment.
See our full guide to leukemia subtypes and treatment differences for more on molecular classification and precision oncology.
Leukemia Type Comparison
| Feature | ALL | AML | CLL | CML |
|---|---|---|---|---|
| Cell origin | Lymphoid | Myeloid | Lymphoid | Myeloid |
| Speed | Acute (rapid) | Acute (rapid) | Chronic (slow) | Chronic (slow) |
| Peak age | Children 2–5 | Adults ≥65 | Adults ≥60 | Adults 40–60 |
| Key marker | BCR-ABL in ~25% adults | FLT3, NPM1, IDH1/2 | Del(17p), TP53, IGHV | BCR-ABL (defining) |
| Primary treatment | Chemo ± TKI | Intensive chemo ± targeted | BTK inhibitors, venetoclax | TKIs (imatinib, etc.) |
| Curative potential | Yes (children >90%) | Yes (with SCT, some subtypes) | Not typically | Yes (TFR or SCT) |
How Leukemia Type Is Diagnosed
Determining the exact leukemia type requires a systematic, multi-step process. The complete blood count (CBC) is the first test to suggest leukemia — but it cannot determine the type definitively. Type determination requires:
- Peripheral blood smear: Morphological assessment of blast cell appearance
- Flow cytometry (immunophenotyping): Identifies the specific surface markers on leukemia cells, confirming lymphoid vs. myeloid origin and B-cell vs. T-cell lineage in ALL
- Bone marrow biopsy: Provides marrow morphology, cellularity, and specimen for cytogenetic/molecular testing. See our full guide to what to expect from bone marrow biopsy.
- Cytogenetics (karyotype and FISH): Identifies chromosomal abnormalities including Philadelphia chromosome, del(17p), trisomies, and translocations
- Molecular testing (PCR and NGS): Detects specific gene mutations (BCR-ABL, FLT3, NPM1, IDH1/2, TP53) that guide targeted treatment selection
The full interpretation of these results is explained in our guide to understanding leukemia test results.
Treatment by Leukemia Type
Treatment is always individualized and type-specific. An overview of available options is provided in our guide to leukemia treatment options. Key type-specific highlights:
- ALL: Intensive multi-agent chemotherapy over 2–3 years; TKIs added for Ph+ ALL; stem cell transplantation for high-risk; CAR-T and blinatumomab for relapsed/refractory disease
- AML: Intensive induction chemotherapy (cytarabine + anthracycline, "7+3"); molecular-targeted agents for specific mutations; allogeneic SCT for eligible high-risk patients; APL treated with ATRA + arsenic trioxide
- CLL: Watch-and-wait for early asymptomatic disease; BTK inhibitors or venetoclax-based therapy for treatment-requiring disease; clinical trials for del(17p)/TP53-mutated high-risk disease
- CML: Daily oral TKI (imatinib first-line, or second/third-generation for resistance); targeted therapy allows most patients to achieve and maintain deep remission
Prognosis Overview by Type
Prognosis in leukemia is type- and subtype-specific. The risk across life stages and molecular profile are the strongest predictors. In general:
- Childhood ALL: Cure rate >90% with modern therapy
- Adult ALL: 5-year survival 35–60%, improving with targeted therapies
- AML: 5-year survival ~29% overall; dramatically better for favorable molecular subtypes (e.g., APL ~90%)
- CLL: Many patients live 10+ years; not generally curable but increasingly controllable as a chronic disease
- CML: Near-normal life expectancy with effective TKI treatment; treatment-free remission achievable for some
Patient Considerations
Patients newly diagnosed with leukemia should seek care at a center with dedicated hematology-oncology expertise. The complexity of leukemia typing — particularly the molecular and cytogenetic analysis — requires specialized laboratory infrastructure and clinical expertise. Second opinions are always appropriate and encouraged for complex cases. Enrollment in clinical trials should be discussed at the time of diagnosis, as many trials offer access to therapies not yet widely available that may improve outcomes.
Caregiver Guidance
Understanding which type of leukemia your family member has — and why that matters for treatment — helps caregivers provide informed support and ask the right questions. Caregivers should not make assumptions about treatment or prognosis based on general "leukemia" information — the differences between types are enormous. Keeping detailed notes at medical appointments, tracking blood test results over time, and maintaining open communication with the oncology team are essential caregiver functions.
When to Seek Urgent Evaluation
If you or a family member has symptoms suggesting leukemia — particularly the cluster of profound fatigue, unexplained bruising, and recurrent infections — seek medical evaluation promptly. A complete blood count is the essential first step. In acute leukemia (ALL or AML), symptoms escalate rapidly and treatment should begin urgently. Do not wait weeks to "see if it resolves." In chronic leukemia, the urgency is lower but still real — earlier diagnosis preserves more options.
🚨 Seek Emergency Care For:
- Fever above 101°F in any suspected or confirmed leukemia patient
- Uncontrolled bleeding from any site
- Sudden breathlessness at rest or rapidly worsening symptoms
- Sudden severe headache or neurological changes
💬 Questions to Ask Your Healthcare Team
- What is the specific type and subtype of my leukemia?
- What molecular markers were found, and how do they affect treatment choices?
- What is the standard first-line treatment for my specific leukemia type?
- Should I be treated at a specialized leukemia center?
- Are there clinical trials I should consider at this stage?
- What is the expected treatment timeline and what does remission mean for my type?
This content is for informational and educational purposes only. It is not a substitute for professional medical advice, diagnosis, or treatment. Always consult your physician or a qualified healthcare provider with any questions about a medical condition. Never disregard professional medical advice or delay seeking it because of information you have read on this website. Read our full disclaimer.
Frequently Asked Questions
The four main types are Acute Lymphoblastic Leukemia (ALL), Acute Myeloid Leukemia (AML), Chronic Lymphocytic Leukemia (CLL), and Chronic Myeloid Leukemia (CML).
In adults, CLL and AML are most common. In children, ALL is by far the most prevalent, accounting for about 75% of pediatric leukemia.
Acute leukemia involves fast-growing, immature cells and progresses rapidly. Chronic leukemia involves mature but abnormal cells that typically progress more slowly.
