📋 Quick Summary
- Topic: The Philadelphia chromosome, the TKI revolution, disease phases, and modern response rates that transformed CML prognosis.
- Key Takeaway: Early recognition and medical consultation are critical for the best clinical outcomes.
- Remember: Always consult a qualified healthcare professional if you have concerns.
Chronic Myeloid Leukemia (CML): Treatment and Outlook
Chronic Myeloid Leukemia (CML) is a myeloproliferative neoplasm defined by the Philadelphia chromosome — a specific chromosomal translocation that creates a constitutively active oncogene. It is one of the most striking success stories in the history of cancer medicine: once a disease with a median survival of only 5–6 years, CML is now managed as a chronic condition with near-normal life expectancy for most patients, thanks to the development of tyrosine kinase inhibitor (TKI) therapy.
CML accounts for approximately 15% of all adult leukemia diagnoses and has an annual incidence of approximately 1–2 cases per 100,000 people. It most commonly affects adults in middle age and beyond, with a peak incidence in the 50–60 age range. It is rare in children but can occur at any age.
CML is characterized by overproduction of myeloid cells (particularly granulocytes) that retain some ability to mature and function, unlike the immature blasts of acute leukemia. This relative preservation of function explains why CML often remains asymptomatic or mildly symptomatic for months to years in its early (chronic) phase before progressing to more aggressive phases.
The Philadelphia Chromosome: CML's Defining Molecular Lesion
The Philadelphia chromosome (Ph) is the product of a reciprocal translocation between chromosomes 9 and 22 — written as t(9;22)(q34;q11). This translocation juxtaposes the BCR gene on chromosome 22 with the ABL1 gene (a tyrosine kinase) from chromosome 9, creating the BCR-ABL1 fusion gene on the shortened chromosome 22 (the Philadelphia chromosome).
The BCR-ABL1 fusion protein is a constitutively active tyrosine kinase — it is permanently switched "on" regardless of normal cellular signals. This drives:
- Uncontrolled proliferation of myeloid cells
- Impaired apoptosis of CML cells
- Altered adhesion to bone marrow stroma (allowing abnormal cell migration)
- Genomic instability (potentially contributing to disease progression)
The BCR-ABL1 fusion gene is the direct drug target of all TKIs used in CML treatment. Imatinib (Gleevec) — the first TKI — was specifically designed to fit into the ATP-binding pocket of the ABL1 kinase domain, blocking its abnormal activity. This was one of the first examples of rational drug design based on a specific oncogenic molecular target, and it launched the targeted therapy revolution in cancer medicine.
The Philadelphia chromosome is also found in ~25–30% of adult ALL — see ALL treatment guide for how TKIs are used in Ph+ ALL.
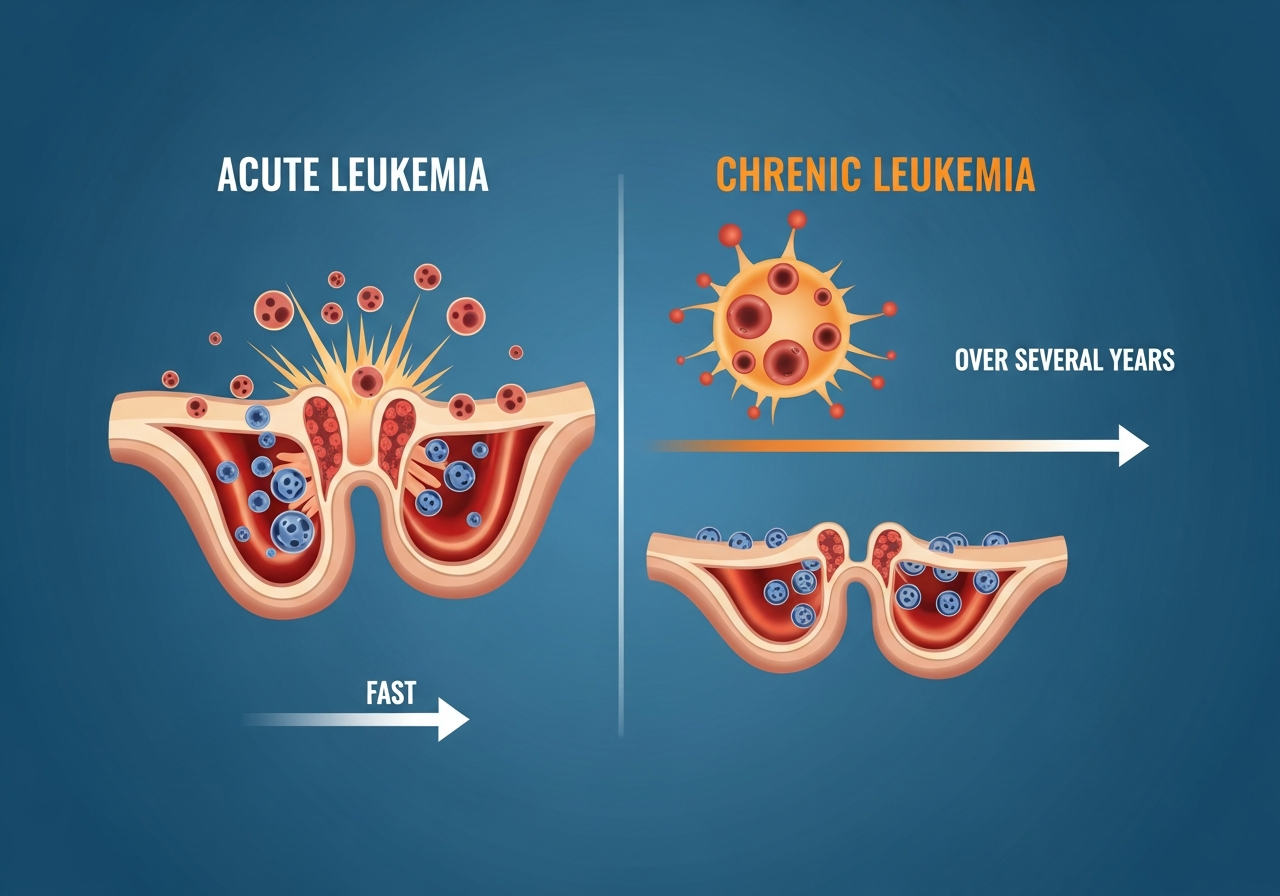
CML Disease Phases
CML progresses through three clinical phases:
Chronic Phase (CP-CML)
The vast majority of patients (85–90%) are diagnosed in chronic phase. Bone marrow blasts are <10%. Symptoms are typically mild — fatigue, splenomegaly-related abdominal fullness, and occasional left shoulder discomfort from diaphragmatic irritation. Many patients are discovered incidentally through a routine blood count showing markedly elevated white blood cells with a full spectrum of myeloid cells (myelocytes, metamyelocytes, band forms, segmented neutrophils, eosinophils, and basophils — a characteristic "left shift" pattern). Chronic phase CML responds very well to TKI therapy.
Accelerated Phase (AP-CML)
Accelerated phase indicates disease progression — defined by one or more of: 15–30% blasts in blood or marrow, ≥20% basophils, persistent thrombocytopenia unrelated to therapy, clonal cytogenetic evolution (new chromosomal abnormalities in addition to the Philadelphia chromosome), or progressive splenomegaly despite treatment. AP-CML requires more intensive management — typically a second or third-generation TKI.
Blast Phase (BC-CML, Blast Crisis)
Blast phase (≥30% blasts in blood or bone marrow, or any extramedullary blast deposit) is clinically equivalent to acute leukemia. It may be myeloid (70%) or lymphoid (30% — Ph+ ALL). Blast crisis has a very poor prognosis with conventional therapy. Treatment includes intensive chemotherapy combined with a TKI, with the goal of returning to chronic phase and proceeding to allogeneic stem cell transplantation.
Symptoms of CML
In chronic phase, CML symptoms are often subtle or absent:
- Fatigue — mild to moderate; related to anemia and high metabolic demand of proliferating cells
- Left upper quadrant pain/fullness from splenomegaly — the most characteristic physical finding; spleen can be massively enlarged at diagnosis
- Left shoulder pain from diaphragmatic irritation by an enlarged spleen
- Unexplained weight loss — modest in chronic phase; more prominent in accelerated/blast phase
- Night sweats — mild to moderate
- Gout or elevated uric acid from high cell turnover
- Priapism (painful prolonged erection) — rare; from leukostasis in genital vasculature
- In blast crisis: severe bone pain, acute fatigue, fever, and bleeding — identical to acute leukemia
Diagnosing CML
CML diagnosis is based on a characteristic CBC pattern combined with confirmation of BCR-ABL1:
- CBC: Markedly elevated WBC (often 50,000–300,000/μL); characteristic myeloid left shift; basophilia and eosinophilia; mildly elevated platelets early; anemia varies. Learn about CBC in leukemia.
- Peripheral blood smear: A characteristic spectrum of myeloid cells at all stages of maturation — essentially the entire granulocyte maturation sequence visible on smear; <10% blasts in chronic phase.
- BCR-ABL1 testing by FISH or RT-PCR: The definitive molecular confirmation. FISH detects the BCR-ABL1 fusion gene in interphase cells. RT-PCR quantifies BCR-ABL1 transcript level — used for diagnosis and for monitoring treatment response.
- Cytogenetics (karyotype): Demonstrates the Philadelphia chromosome — t(9;22) — in 95% of CML; the remaining 5% have "cryptic" translocations detectable only by FISH or PCR.
- Bone marrow biopsy: Confirms phase (blast percentage) and evaluates bone marrow architecture. About bone marrow biopsy.
Tyrosine Kinase Inhibitors: The Revolution in CML Treatment
TKIs are the standard treatment for chronic phase CML and have transformed the disease from a uniformly fatal malignancy to a manageable chronic condition for the vast majority of patients.
First-Line TKI Options
- Imatinib (Gleevec/Glivec): The original TKI; FDA-approved in 2001; daily oral dosing (400 mg); well-tolerated; now off-patent with generic availability. At 10-year follow-up in the IRIS trial, 83% of patients remained in complete cytogenetic remission. Now considered less potent than second-generation agents but well established with long safety data.
- Nilotinib (Tasigna): Second-generation TKI; more potent than imatinib; higher rates of major molecular response at 12 months; associated with cardiovascular risk (peripheral arterial occlusive disease) with prolonged use — requires cardiovascular monitoring.
- Dasatinib (Sprycel): Second-generation TKI; more potent than imatinib; penetrates CNS (relevant for blast phase); associated with pleural effusion; once-daily dosing.
- Bosutinib (Bosulif): Second/third-generation; used in first line or after resistance/intolerance; primarily GI side effects.
- Ponatinib (Iclusig): Third-generation TKI; active against the T315I "gatekeeper" mutation that confers resistance to all other TKIs; used for resistant/intolerant disease and T315I-positive CML; significant cardiovascular and vascular risk requiring careful monitoring.
- Asciminib (Scemblix): A STAMP (Specifically Targeting the ABL Myristoyl Pocket) inhibitor — a completely different binding mechanism from ATP-competitive TKIs; highly active against T315I mutation at high dose; FDA-approved for third-line+ treatment.
Monitoring Treatment Response in CML
Treatment response in CML is monitored by serial quantitative PCR (RT-qPCR) measuring BCR-ABL1 transcript levels in peripheral blood:
- Complete Hematologic Response (CHR): Normal blood counts — typically achieved within 3 months
- Complete Cytogenetic Response (CCyR): No Ph+ cells detectable by karyotype — typically achieved by 12 months
- Major Molecular Response (MMR / MR3.0): BCR-ABL1/ABL1 ratio ≤0.1% on the International Scale — the primary treatment goal in standard management; typically achieved by 12–18 months
- Deep Molecular Response (MR4.0 / MR4.5): BCR-ABL1 ≤0.01%–0.0032% — the threshold required for attempted treatment-free remission
Suboptimal responses at specific time points (e.g., BCR-ABL1 >10% at 6 months, >1% at 12 months) are "warning" or "failure" signals that may warrant TKI switch or dose escalation. Serial monitoring is typically performed every 3 months during the first 2 years, then every 3–6 months. See understanding leukemia test results for more on molecular response monitoring.
Treatment-Free Remission (TFR): Can Patients Stop Their TKI?
One of the most important developments in CML management in the past decade is the discovery that a substantial minority of patients who have achieved and maintained deep molecular remission (MR4.5 sustained for at least 2 years) can discontinue TKI therapy and maintain remission without any treatment — so-called Treatment-Free Remission (TFR).
Approximately 40–60% of patients who attempt TKI discontinuation after achieving sustained deep molecular remission maintain TFR at 12 months. The remainder experience "molecular relapse" — a rise in BCR-ABL1 levels — typically within the first 6 months. Molecular relapse is not blast crisis — it is detected by PCR monitoring before any clinical symptoms develop. Restarting the same TKI at the same dose rapidly brings BCR-ABL1 levels back under control in virtually all patients who relapsed during TFR.
TFR attempts should only be made at specialized CML centers with access to frequent molecular monitoring. Patients who succeed in maintaining TFR have quality of life equivalent to the general population, free from daily medication and its side effects.
Prognosis in CML
The prognosis for CML in chronic phase treated with modern TKIs is excellent. Major registry data show that CML patients treated with imatinib have overall survival rates approaching those of age-matched general populations — a remarkable transformation from the pre-TKI era. Second-generation TKIs produce faster and deeper molecular responses, and their long-term survival data are also excellent.
Factors associated with better outcomes include: early diagnosis in chronic phase, prompt achievement of optimal molecular milestones (MMR at 12 months), absence of high-risk Sokal/EUTOS score features, and achievement of sustained deep molecular remission. Patients who progress to accelerated or blast phase have significantly worse prognoses, underscoring the importance of adherence to TKI therapy and regular monitoring.
Living with CML: Adherence and Quality of Life
CML is a disease patients manage for many years — potentially decades. The most critical patient responsibility is TKI adherence: missing doses, particularly more than 25% of doses, is strongly associated with failure to achieve or maintain molecular response milestones. Patients should develop a routine for taking their TKI and understand that subtherapeutic exposure can lead to treatment resistance.
Common TKI side effects that affect quality of life include muscle cramps, fluid retention, nausea, rash, fatigue, and — for second-generation agents — cardiovascular effects. Proactive side effect management with the treatment team is important to maintain adherence. Learn more about targeted therapy for leukemia.
Fertility considerations are important for younger patients. Imatinib and other TKIs can affect fertility and should not be taken during pregnancy. Reproductive planning should be discussed explicitly before starting treatment. For women in remission who wish to conceive, TKI discontinuation under specialist supervision during pregnancy monitoring is feasible with careful management.
Caregiver Guidance
CML caregivers often have a unique role — supporting a patient who appears relatively well (particularly in early chronic phase) but who requires indefinite daily oral medication and regular monitoring. Caregivers can help by supporting adherence (reminders, organizing medications), attending molecular monitoring appointments, and being alert to changes in symptoms that may indicate disease progression (escalating fatigue, worsening splenomegaly, fever, bone pain). In the event of blast crisis — which can occur rapidly — caregiver recognition of the dramatic symptom change and immediate medical contact is essential.
When to Seek Medical Attention in CML
- Fever above 101°F — infection in CML patients with neutropenia (during accelerated/blast phase) is serious
- Sudden severe left-sided abdominal pain — possible splenic infarction or rupture from massive splenomegaly
- Rapid onset of AML-like symptoms (severe fatigue, bruising, bleeding, very high WBC at any count) — possible blast phase transition
- New BCR-ABL1 rise after previous deep molecular response — requires same-day contact with oncology team, not emergency room (unless symptomatic)
- Signs of vascular events on TKI therapy (chest pain, leg pain, peripheral coldness) — TKI-related cardiovascular toxicity
🚨 When CML Patients Need Urgent Evaluation
- Acute AML-like symptoms (rapid onset bruising, bleeding, fever) — possible blast crisis
- Sudden severe abdominal pain with known large spleen — possible splenic event
- Chest pain, acute leg pain, or vascular symptoms on TKI — cardiovascular emergency
💬 Questions to Ask Your CML Treatment Team
- What TKI should I start, and why this choice for my specific situation?
- What molecular response milestones should I be achieving at 3, 6, and 12 months?
- How often will my BCR-ABL1 levels be monitored?
- Am I ever eligible to try stopping my TKI (treatment-free remission)?
- What are the cardiovascular risks of my specific TKI and how will they be monitored?
- What changes in my symptoms should prompt me to contact you immediately?
This content is for informational and educational purposes only. It is not a substitute for professional medical advice, diagnosis, or treatment. Always consult your physician or a qualified healthcare provider with any questions about a medical condition. Never disregard professional medical advice or delay seeking it because of information you have read on this website. Read our full disclaimer.
Frequently Asked Questions
A genetic abnormality created by a translocation between chromosomes 9 and 22, producing the BCR-ABL fusion gene that drives CML.
TKIs like imatinib (Gleevec) have transformed CML from a fatal disease to a manageable condition, with most patients achieving deep molecular remission.
While TKIs control CML excellently, a true cure typically requires stem cell transplantation. Some patients achieve treatment-free remission on TKIs.
